|
The standard therefore recommends that you first determine the impact of the process on the QMS’ effectiveness and product conformity, before deciding how to measure or monitor. In ISO 9001, it is mentioned that attempting to measure and monitor everything is expensive and, in most cases, virtually impossible. In this is not done, effectiveness can never be achieved. If you for example monitor the corrective actions process, and you plan not to have any problems repeating once corrective actions have been declared effective, and various problems still repeat, the problem’s root cause needs to be first identified and then addressed. If the results do not align with the objectives, the process should be corrected. It is crucial to compare the results of a process to the results that are expected and planned for as defined by the objectives of the process. This is after all the purpose of implementing a QMS in the first place. Step one to improve any process is to make sure that the process behaves as planned. Deviations may only be permitted when an authorized company representative and / or the customer has approved this in writing.Īll QMS processes have to be measured where possible, and always monitored to make sure that they achieve what they were designed to do. The records that approve the release should also indicate by whom the release was authorized. These planned criteria are sometimes known as the product’s quality plan. A product should for example include any test requirement, requirements determining weight, size and any other measurements, and occasionally the environment in which the product must be created, for example the cleanliness and hygienic requirements needed in food production.īefore the service or product is finally released, it should meet all the planned criteria. The planned arrangement for services or products comprises all the acceptance requirements and the making of the service or product. Records should always be maintained to show that the product conforms to the criteria for acceptance. The crucial reason for monitoring and measuring products is to confirm its characteristics against planned requirements at various process stages. Step one in achieving customer satisfaction is ensuring that the service or product meets their requirements, the product monitoring and measuring requirements are very important. The standard requires that measurement and monitoring be used in a number of different places. Although both are meaningful tools, they do in fact provide different types of evidence that can be used in decision making. sizes, volume, etc.) Monitoring would therefore be used to determine when a process approaches a limit, while measuring would give you the real value of parameters being looked at and determine how it is different from the value that is required. On the other hand, measurement is to determine the actual characteristics of something (e.g. We monitor by observing something closely so that you can record or detect. The difference between Measurement and monitoringĪlthough the words measurement and monitoring are always used together right through ISO 9001, they mean different things. Monitoring and measuring provides you with this evidence. 
To use this method, you should gather evidence that can be used for decision making. This will clearly show you how improvements are going. Data gathered is used to look at the actual result of the process and this is then compared to the intended result that was defined in the quality objective for the process. ISO 9001 is based on a major principles of any decent QMS, which is decision making based on evidence. 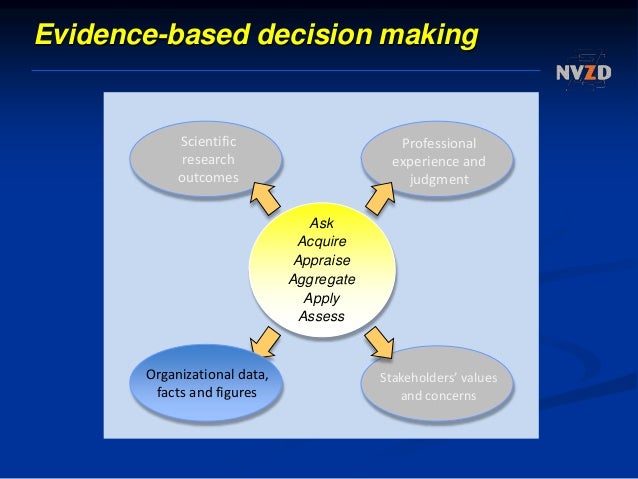
A big part of ISO 9001 deals with measurement and monitoring and this makes sense.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
